Purdue Pharma L.P. Receives FDA Approval for 7.5 mcg/hour Dosage Strength of Butrans® (Buprenorphine) Transdermal System CIII
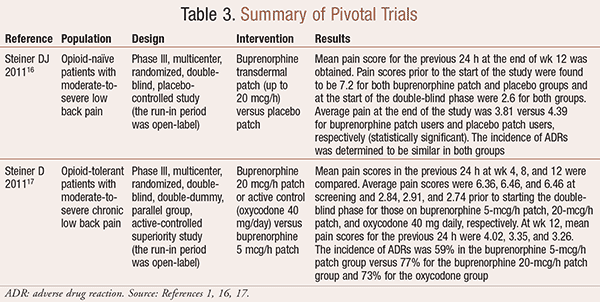
Title: Analysis of abuse of buprenorphine transdermal delivery system ( Butrans®) using data from the RADARS® System Poison Cen
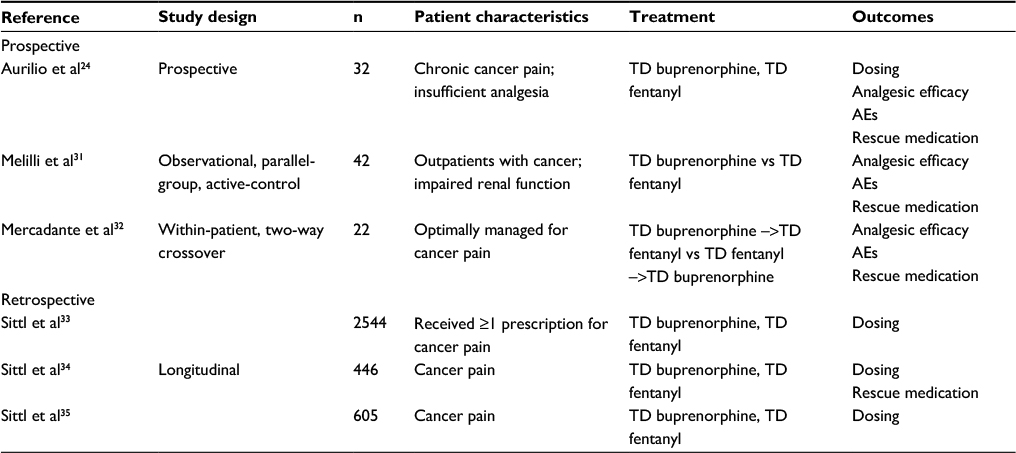
Full article: Comparison of abuse, suspected suicidal intent, and fatalities related to the 7-day buprenorphine transdermal patch versus other opioid analgesics in the National Poison Data System

Butrans® (buprenorphine) Transdermal System CIII | What is Butrans® ( buprenorphine) Transdermal System CIII?